- Home
- BLOG & STORIES
BLOG & STORIES
Introducing A Decade of Clarity
by Denise Purdie Andrews | May 6, 2026 | Announcements

In 2026, Axis Forensic Toxicology marks ten years as Axis.
To recognize that milestone, we are proud to introduce our anniversary theme: A Decade of Clarity.
This theme reflects more than time passed. It reflects ten years of building, evolving, and investing in the science, service, and relationships that matter most to the forensic community.
Since 2016, Axis has remained focused on delivering clear answers in complex cases, responsive support, and continued investment in the people, systems, and capabilities that strengthen our work.
As we move through 2026, A Decade of Clarity will serve as a simple expression of what this milestone means to Axis: not just ten years in business, but ten years of steady commitment to clarity, partnership, and progress.
We are grateful for the opportunity to serve the forensic community and thankful for the clients and partners who have been part of this journey.
Thank you for being part of the Axis story.
Phil Roberts
Chief Executive Officer
Axis Forensic Toxicology
Emerging Substances – The “Orphines” Class of Designer Opioids
by Denise Purdie Andrews | Apr 21, 2026 | Drug Classes
By Kevin G. Shanks, D-ABFT-FT
The term designer opioid generally refers to the class of synthetically derived opioid analogs that are intentionally modified by clandestine chemists to circumvent existing drug laws while maintaining opioid receptor activity. This concept has been ongoing over the years with the initial appearance of fentanyl analogs such as alpha-methylfentanyl and 3-methylfentanyl during the 1980s and the reemergence of them on illicit drug markets in the 2010s. Shortly thereafter, the fentanyl class of compounds was heavily controlled by the government, and clandestine labs moved on to the synthesis of the nitazene family of opioids, which gained prominence in the late 2010s to early 2020s. As the nitazenes have become regulated, the market looks to be shifting again, and the orphine class of opioids has the potential to be the next type of designer opioid to become prevalent.
The orphine class of substances have an origin in academic and pharmaceutical research conducted during the 1960s and 1970s, particularly in laboratories associated with Paul Janssen, whose work also produced fentanyl and related fentanyl analogs. Many of these compounds were initially synthesized as potential analgesics, but they remained relatively obscure within scientific literature and patents. In the last few years, these compounds have been rediscovered and have found their way to the street as illicit drugs.
The modern recognition of the orphine class began with the detection of brorphine on illicit drug markets in Europe in 2019 and in the United States in 2020. Brorphine is a benzmidazol-2-one derivative that contains a cyclized benzimidazole ring system. Other compounds in this class include cyclorphine (also known as N-propionitrile chlorphine), and 5,6-dichloro desmethylchlorpine (also known as S-17018), and 5,6-dichloro brorphine (also known as SR-14968).
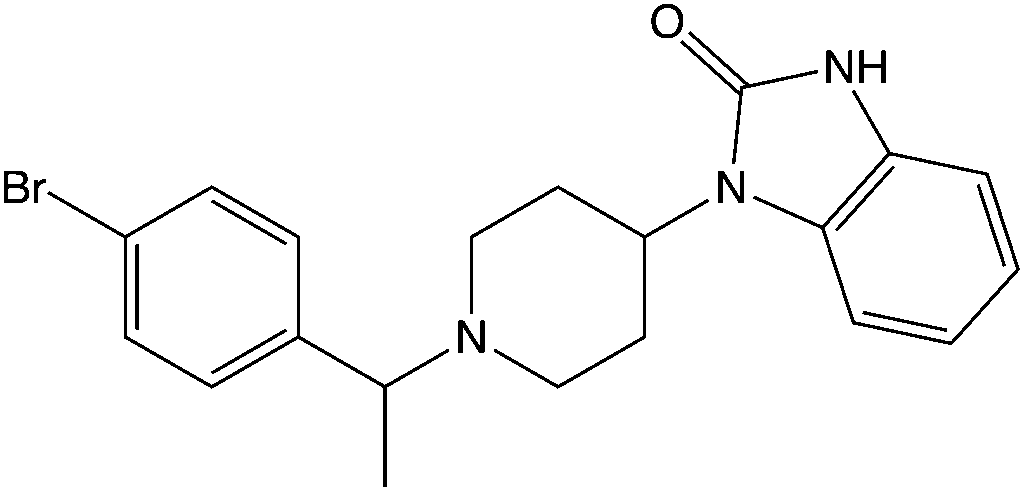
Chemical Structure of Brorphine Drawn by Kevin G. Shanks (2026)
The other structural group of orphine compounds are the spiropiperidine derivatives, which contain a spiro-linked piperidine ring fused with an imidazole or other related aromatic system. Compounds in this class include spirorphine, spirochlorphine, and spirobrorphine.
The orphines may differ from the traditional morphine derivatives and the typical fentanyl analogs in chemical structure, but the pharmacological effect is similar, as they act as potent mu opioid receptor agonists. This activity produces analgesia, euphoria, sedation, central nervous system depression, and respiratory depression. In overdose, the breathing slows down, and may stop temporarily (apnea). Apnea leads to hypoxia, or lack of oxygen distribution to the surrounding tissues including the brain. Hypoxia can lead to cardiac arrest and death.
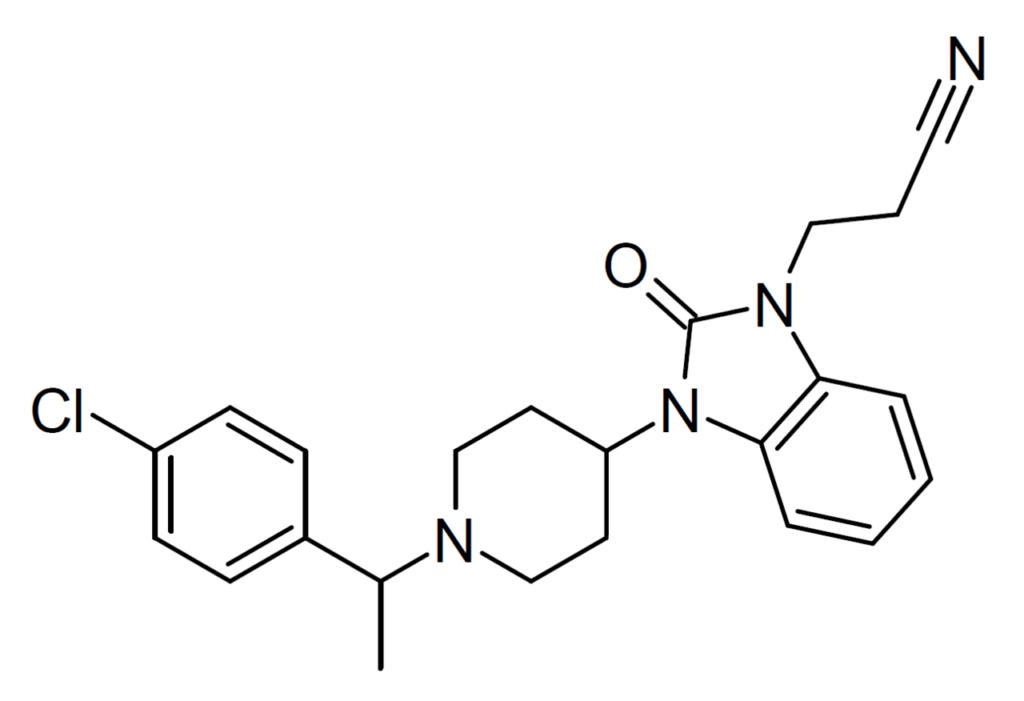
Chemical Structure of Cyclorphine Drawn by Kevin G. Shanks (2026)
The orphine class of opioids is particularly concerning from a toxicological perspective as some of the analogs are considered to be high potency, with several of them being similar to fentanyl in pharmacological activity. Many of these compounds have very limited toxicological data as they never underwent routine animal or human clinical testing. Because of these factors and with the potential increased prevalence in drug materials on the street, forensic toxicology laboratories have begun expanding their analytical methods to identify these compounds in postmortem analyses. As always, the cycle of innovation by clandestine chemists and then governmental prohibition drives the continual appearance of new opioids on the drug market. Although only a limited number of analogs have been detected so far, their appearance illustrates how the drug market changes rapidly over the course of months and years.
Axis Forensic Toxicology has been testing for brorphine since 2023 and recently added chlorphine, N-propionitrile chlorphine (cyclorphine), 5,6-dichloro brorphine, 5,6-dichloro desmethylchlorphine, spirobrorphine, and spirochlorphine to a newly developed Orphine Analog Panel (order code 13410), and Comprehensive Panel, Blood with Analyte Assurance™ (order code 70510) using liquid chromatography with quadrupole time of flight mass spectrometry (LC-QToF-MS). Reporting of these analytes is qualitative. Reporting limits are 10 ng/mL for 5,6-dichloro brorphine and 5,6-dichloro desmethylchlorphine and 1 ng/mL for chlorphine, N-propionitrile chlorphine, spirobrorphine, and spirorchlorphine.
If you have any questions or concerns regarding the role of these orphine opioids or any other newly emerged substance in your investigation, please reach out to our Axis Forensic Toxicology subject matter experts at [email protected] or by phone (317-759-4869, Option 3). To stay current with the scope of testing for all services offered by Axis, please consult the online catalog.
Read MoreUpcoming Additions to Comprehensive Panel with Analyte Assurance™ and New Orphine Analog Panel
by Denise Purdie Andrews | Apr 16, 2026 | Announcements
We’re pleased to inform you that effective April 20th, 2026, Axis will be updating our Comprehensive Panel with Analyte Assurance™ and offering a new Orphine Analog Panel.
What is Changing?
For orders placed on or after April 20th, 2026, the 70510 Comprehensive Panel, Blood will include the following added analytes:
- alpha-PHPP (PV8) – Confirmed via 13710: Novel Emerging Compounds Panel
- alpha-PiHPP (Iso-PV8) – Confirmed via 13710: Novel Emerging Compounds Panel
- alpha-PipHP – Confirmed via 13710: Novel Emerging Compounds Panel
- Ethylbromazolam – Confirmed via 13510: Designer Benzodiazepine Panel
- Mitragynine Pseudoindoxyl – Confirmed via 13710: Novel Emerging Compounds Panel
- N-Pyrrolidino Ethylene Isotonitazene – Confirmed via 13910: Nitazene Analog Panel
- N-Pyrrolidino Isotonitazene – Confirmed via 13910: Nitazene Analog Panel
- Phenazolam – Confirmed via 13510: Designer Benzodiazepine Panel
- Suzetrigine – Provided as part of 70510: Comprehensive Panel, Blood
Additionally, with the rise of “Orphines” throughout the United States, Axis will be offering a new 13410: Orphine Analog Panel, Blood. As with other emerging and designer type panels, the screening for these analytes will be included as part of Analyte Assurance in the 70510: Comprehensive Panel, Blood. The components of this panel are detailed below.
- 5,6-Dichloro Brorphine
- 5,6-Dichloro Desmethylchlorphine
- Brorphine
- Chlorphine
- N-Propionitrile Chlorphine (Cychlorphine)
- Spirobrorphine
- Spirochlorphine
What Do You Need to Do?
You do not need to do anything different as these analytes will automatically be included with your 70510: Comprehensive Panel, Blood order under Analyte Assurance. For a copy of the new specification sheet that is effective April 20th, 2026 please click this link: https://axisfortox.com/test_catalog/comprehensive-drug-panel/
At Axis Forensic Toxicology, we are committed to evolving with the needs of our clients and the ever-changing landscape of emerging substances. These additions reflect our proactive approach to ensuring you receive the most comprehensive and reliable testing services available.
If you have any questions or would like more information regarding this update, please don’t hesitate to reach out to us at [email protected]. Our team is always ready to assist.
Thank you for your continued partnership and trust in Axis. We look forward to continuing to support your work with the highest quality service and testing solutions.
Sincerely,
Matt Zollman
Director of Operations & Product Management
Axis Collaborates with Forensic and Investigative Sciences Program
by Denise Purdie Andrews | Mar 12, 2026 | Uncategorized
Axis is fortunate to have strong university science programs within the state of Indiana to supply forensic scientists to work in its laboratory. Axis maintains a particularly strong relationship with the Forensic and Investigative Sciences (FIS) program at Indiana University Indianapolis (formerly Indiana University Purdue University Indianapolis). Axis’ Chief Operating Officer, Denise Purdie Andrews, serves on the FIS Board of Visitors, and Axis’ Director of Operations and Product Management, Matt Zollman, recently served on a panel presentation associated with the recent FIS 20th Anniversary Symposium.
Axis’ longest-standing relationship is with toxicologist Kevin Shanks, who has served from time to time as a speaker and instructor over the past decade. Kevin has also collaborated with its faculty and students on research. This research has culminated with a published paper, “Paper Spray Mass Spectrometry for Rapid Drug and Drug Metabolite Screening Directly from Postmortem Blood Samples”. It is available for review here: https://www.ojp.gov/library/publications/paper-spray-mass-spectrometry-rapid-drug-and-drug-metabolite-screening
Axis is pleased to engage with others in the forensic science community and to support the research efforts of students and faculty in Indiana and beyond.
Read MoreAxis Experts Present at MATT 2026
by Denise Purdie Andrews | Mar 2, 2026 | Announcements
Axis toxicologists Laureen Marinetti, PhD, F-ABFT, and Stuart Kurtz, M.S, D-ABFT-FT, will be presenting at the Midwest Association of Toxicology and Therapeutic Drug Monitoring (MATT) Annual Meeting in Cincinnati, Ohio, March 18-20, 2026. Their presentation topics are below and the presenter is highlighted.
Axis is pleased to be able to share knowledge with other professionals in the fields of toxicology. If you will be attending MATT, please make plans to connect with them and take in their presentations.
Read MoreAxis Offers Training Opportunities for Forensic Pathology Medicine Fellows
by Denise Purdie Andrews | Feb 19, 2026 | General
By Kevin G. Shanks, M.S., D-ABFT-FT
Axis Forensic Toxicology prides itself in its ability and willingness to provide continuing education and training for our forensic toxicology clients. One of these educational avenues is the Axis forensic medicine fellow training rotation program. We are planning now for our Spring 2026 program.
In this weeklong virtual training program, a Forensic Medicine Fellow will be introduced to the set-up and operation of the modern postmortem forensic toxicology laboratory, alongside descriptions of the analytical instrumentation and analytical methods employed by the laboratory. Board certified forensic toxicologists will discuss the importance of toxicology specimen collection and present a survey of the major illicit and pharmaceutical drugs (e.g. ethanol, cocaine, methamphetamine, MDMA, heroin, fentanyl, and prescription opioids) with respect to mechanisms of actions, their detection, and their relevance to cause of death determination. Novel psychoactive substances (NPS), to include designer benzodiazepines, synthetic cannabinoids, substituted cathinones, fentanyl analogs, nitazene derivatives, xylazine, and mitragynine (kratom) are also discussed in depth.
The forensic toxicology forensic medicine fellow training rotation instills the Fellow with confidence in interpreting the significance of toxicological findings with respect to cause and manner of death classification. Peer reviewed scientific references provided to the Fellow lay a foundation for a body of knowledge that may aid in the resolution of drug-related deaths by the forensic pathologist. One-on-one toxicology case review with a forensic toxicologist surveying completed cases by the laboratory will be undertaken and is intended to give the Fellow an overview of how a “toxicology pending conference” is conducted between forensic pathologists and forensic toxicologists.
If you are a forensic pathology fellow or a pathologist who directs forensic pathology fellows and are interested in learning more about this training opportunity, please reach out to Axis Forensic Toxicology’s toxicologists at [email protected] or call us on the phone at (317) 759 – 4869.
Read MoreDrug Primer: BTMPS
by Denise Purdie Andrews | Feb 6, 2026 | Drug Classes
By Laureen J. Marinetti, PhD, F-ABFT
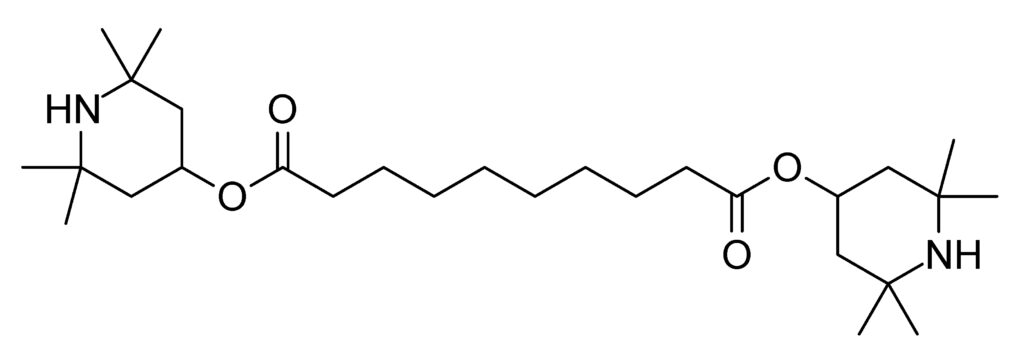
BTMPS (Bis[2,2,6,6-tetramethyl-4-piperidyl] sebacate), brand name Tinuvin®770, is commonly used in the manufacture of plastics as a UV-stabilizing additive to prevent degradation from sunlight and weather exposure. BTMPS was first identified in the U.S. illicit drug market in mid-2024 and spread rapidly across the country. The first identifications were by community drug checking organizations which identified BTMPS using Fourier Transform Infrared spectroscopy (FTIR). BTMPS is not the only impurity that may be found in the drug supply about which little is known, and not even the only light stabilizer normally used in manufacturing. For example, 2,2,6,6-tetramethyl-4-piperidinol has also been detected, but not in the quantities observed for BTMPS.
BTMPS is often found in samples alongside fentanyl, synthetic opioids, and stimulants, and sometimes at a greater concentration than the illicit drug. BTMPS’s appearance coincided with disruptions in the usual drug supply chain and reports of unusual odors and tastes in samples. BTMPS does not produce a “high” or any known psychoactive effects in humans. It may be used as a bulking agent to increase volume for profit or potentially to stabilize other chemical precursors.
BTMPS is not approved or studied for human use. Animal studies suggest potential cardiotoxicity, ocular damage, neurotoxicity, and sudden death. The safety data sheet for BTMPS states that human exposure can result in eye damage and skin irritation. BTMPS is a potent calcium channel blocker and nicotine receptor antagonist, which may enhance the toxicity of cooccurring substances.
Axis has confirmed one positive case with BTMPS in a blood sample. The case was an oxycodone overdose with a very high concentration of oxycodone and no illicit drugs detected. It is possible that the BTMPS was a contaminant on the oxycodone pills from the plastic medication bottle, and because so many of them were ingested at one time, the BTMPS accumulated to a detectable concentration. In the Washington drug detection data, it was found that in the following potential licit drug samples, BTMPS was detected. In 296 prescription opioid samples, 3.4% were positive for BTMPS, and in 163 samples sold as M30s (prescription oxycodone) or Percocet, 3.7% were positive for BTMPS.
BTMPS likely ended up in the illicit drug supply because it is cheap, readily available, and possibly introduced through contaminated precursor chemicals or manufacturing streams. It provides no known benefit to users and may increase health risks due to its toxicological profile. Axis monitors BTMPS in the Novel Emerging Compounds (NEC) panel (order code 13710) and Comprehensive Panel, Blood with Analyte Assurance™ (order code 70510) using liquid chromatography with quadrupole time of flight mass spectrometry (LC-QToF-MS).
If you have questions about BTMPS and how it may play a role in your medical-legal investigation, please reach out to Axis subject matter experts by email ([email protected]) or phone (317-759-4869, Option 3).
References
- UV Stabilizer BTMPS in the Illicit Fentanyl Supply in 9 US Locations, Letter, Journal of the American Medical Association, February 5, 2025.
- Where do we see BTMPS?, Addictions, Drug and Alcohol Institute, University of Washington, https://adai.washington.edu/WAdata/DrugChecking/BTMPS.html, 2025.
Drug Primer: MDPHP
by Denise Purdie Andrews | Jan 19, 2026 | Drug Classes
By Kevin Shanks, D-ABFT-FT
Stimulants come and go. New ones arrive on the illicit drug market all the time. One of the newest stimulants to be identified is MDPHP, also known as 3′,4′-Methylenedioxy-α-pyrrolidinohexiophenone. It is a substituted cathinone, a class of compounds related to cathinone, a naturally occurring stimulant alkaloid found in the plant Catha edulus (khat). MDPHP belongs to the pyrrolidinophenone subclass and is structurally related to older stimulant compounds, alpha-PVP and MDPV. MDPHP first appeared on world drug markets around 2014-2015, but has recently seen an increase in prevalence. While not explicitly listed as a controlled substance in the United States, it may be considered a positional isomer of the already controlled MDPV and alpha-PVP, and therefore be considered illegal to manufacture, possess, distribute, and consume.
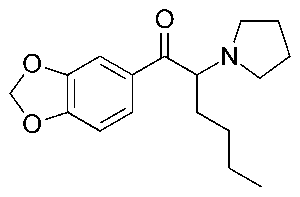
Chemical Structure of MDPHP drawn by Kevin G. Shanks (2026)
Pharmacologically, the substituted cathinones class primarily acts on monoamine transporters in the body and inhibits the reuptake of dopamine, norepinephrine, and/or serotonin and may act as releasers of any of the three neurotransmitters. MDPHP acts primarily as a dopamine and norepinephrine reuptake inhibitor. The end result of this action is an increased concentration of neurotransmitters in the nerve cell synapse, which leads to stimulant physiological effects on the body. People who use stimulants are typically seeking the desired effects such as euphoria, increased energy, sociability, and alertness. But adverse effects include anxiety, agitation, paranoia, hallucinations, and aggression. When taken in overdose, acute toxicity is demonstrated via hypertension, hyperthermia, tachycardia, severe agitation, hallucinations, seizure, kidney failure, rhabdomyolysis, and death. Chronic use can manifest in cognitive impairment, sleepiness/tiredness, psychosis, physical and psychological dependence and addiction.
MDPHP has been implicated in fatalities in peer-reviewed published literature. In one case published by Di Candia et al., a 48 year old male with back and leg pain was found semi-unconscious on a public street. Emergency personnel were called and they documented history to include an HIV+ status and amnesia. After arrival at the hospital, his breathing and heart rate increased and he became unconscious before respiratory and cardiac activity stopped and could not be regained. At autopsy, various bone fractures, cerebral and pulmonary congestion and edema, and atrial and ventricular hemorrhages were documented. Toxicological analysis of postmortem femoral blood was positive for MDPHP (399 ng/mL).
In a second case published by Croce et al. a 30 year old male with a history of drug use and addiction was found deceased in his bedroom. He was last seen alive 24 hours prior to discovery. At autopsy, pleural visceral congestion, pulmonary edema, and moderate hepatic steatosis were observed. Toxicological analysis was completed on postmortem central blood and peripheral blood, among other specimens drawn at autopsy. MDPHP was positive in the central blood (1,639.99 ng/mL) and in the peripheral blood (1,601.90 ng/mL).
In a third case published by Casati et al. a 58 year old male was found floating in the water. He was last seen alive 7 days prior. Documented medical history included HIV+ status and he regularly used MDPV. At autopsy, rigor was present. Both hands were macerated and the heart was enlarged with mild ventricular hypertrophy. Cerebral and pulmonary congestion were also observed. Toxicological analysis was positive for MDPHP in femoral blood (350 ng/mL) and central blood (110 ng/mL). Other substances present included MDPV, MDPPP, MDPBP, citalopram, clonazepam, and 7-aminoclonazepam.
As a new stimulant, it is prudent to be aware of MDPHP and its availability on the illicit drug market. Axis monitors MDPHP in the Novel Emerging Compounds (NEC) panel (order code 13710) and Comprehensive Panel, Blood with Analyte Assurance™ (order code 70510) using liquid chromatography with quadrupole time of flight mass spectrometry (LC-QToF-MS).
If you have questions about MDPHP and how it may play a role in your medical-legal investigation, please reach out to subject matter experts by email ([email protected]) or phone (317-759-4869, Option 3).
References
- Di Candia, D., Boracchi, M., Ciprandi, B. et al. (2022) A unique case of death by MDPHP with no other co-ingestion: a forensic toxicology case. International Journal of Legal Medicine. DOI: https://doi.org/10.1007/s00414-022-02799-w
- Croce, E.B., Dimitrova, A., Grazia Di Mili, M. et al. (2025) Postmortem distribution of MDPHP in a fatal intoxication case. Journal of Analytical Toxicology. DOI: https://doi.org/10.1093/jat/bkae092
- Casati, S., Ravelli, A., Dei Cas, M. et al. (2025) Polydrug fatal intoxication involving MDPHP: Detection and in silico investigation of multiple 3,4-methylenedioxy-derived designer drugs and their metabolites. Journal of Analytical Toxicology. DOI: https://doi.org/10.1093/jat/bkaf048
Drug Primer: Diphenidine
by Denise Purdie Andrews | Jan 9, 2026 | Drug Classes
By Stuart Kurtz, D-ABFT-FT
Drug Primer: Diphenidine
1-(1,2-diphenylethyl)piperidine, commonly known as diphenidine, is part of the 1,2-diarylethylamine class. This class includes drugs such as phencyclidine (PCP), 3-methoxyphencyclidine (3-MeO-PCP), and ketamine. These compounds can interact with many different systems in the body depending on the groups attached to the core structure. These pharmacological interactions can include activation of opioid receptors, inhibition of monoamine transporters, and antagonism of glutamatergic N-methyl-D-aspartate (NMDA) receptors. NMDA receptor antagonism is part of the dissociative aspect of these compounds but how they affect other receptors is also a factor.
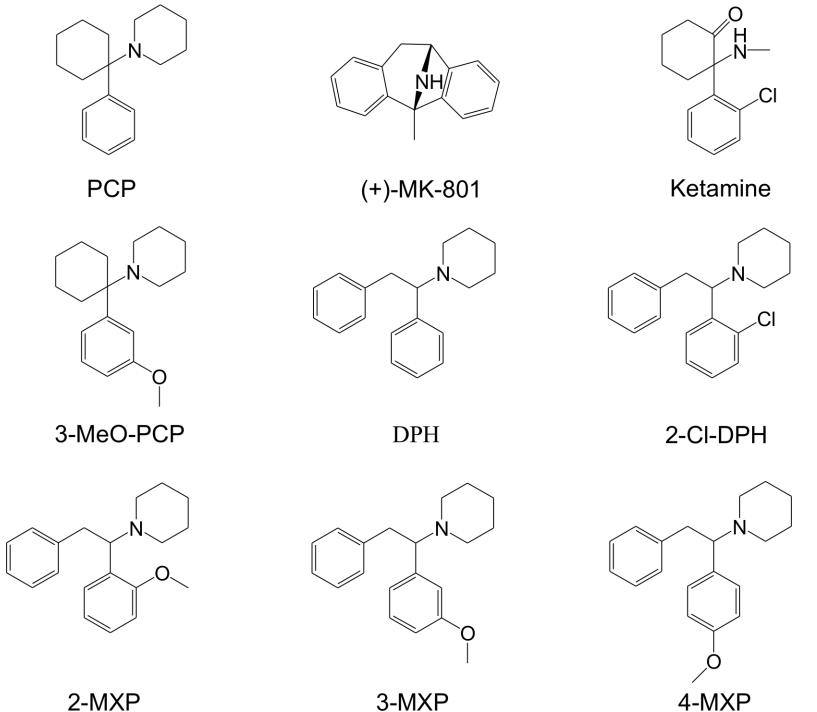
Structures of several compounds related to PCP. Diphenidine is pictured in the bottom row in the middle. From Wallach, et. al., 2016.
Diphenidine interacts with NMDA receptors, serotonin transporter inhibitors, dopamine receptors, and opioid receptors. There have been no human clinical trials for diphenidine so the effects in humans are not well known. Similarity to other substances, such as ketamine, can give some estimation of its effects. Limited toxicity data is available from documented cases of exposure that can also help determine what effects can be expected. Dissociative effects have been reported at lower doses with higher doses causing somatosensory phenomena and transient anterograde amnesia. Reported duration of these effects is about 3-6 hours. Reported adverse effects include hypertension, tachycardia, agitation, muscle rigidity, anxiety, confusion, disorientation, dissociation, and hallucinations.
In one case report, a 30 year-old white male was found confused and agitated by his bed. A small baggie labeled as 1g diphenidine was on the floor nearby. Midazolam was administered by first responders at the scene and on route to the hospital with minimal effect on his mental state. Observed symptoms included increased heart rate (tachycardia), increased respiratory rate (tachypnea), and small (miotic) pupils. Examination at the hospital determined body temperature was slightly elevated at 100.4°F (38.0°C) and metabolic acidosis with a blood pH of 7.17. Normal blood pH is 7.35-7.45. Midazolam, diazepam, chlorpheniramine, haloperidol, and sodium bicarbonate were administered intravenously to sedate him and after 90 minutes, he regained consciousness and did not experience any amnesia. He reported to have taken diphenidine approximately 5 hours prior to being found by first responders. After 12 hours, he was discharged with stable heart rate, stable blood pressure, and normal body temperature.
Diphenidine is not currently scheduled in the United States by the DEA. It is typically sold as a white or yellowish-white powder and consumed by snorting or oral ingestion. It’s marketed as a research chemical and does not have any medical or veterinary use. Because diphenidine exists in a legal gray area, there may be a traceable paper trail, such as records from online vendors and/or bank states documenting its purchase. Additionally, the packaging that it arrives in may be labeled with identifying information.
Axis now screens and confirms for Diphenidine in the 70510 Comprehensive Panel, Blood and Analyte Assurance. For questions about diphenidine or other assistance with interpretation, please call us at 317-759-4869 option 3 or email us at [email protected].
- Wallach J, Kang H, Colestock T, Morris H, Bortolotto ZA, et al. (2016) Pharmacological Investigations of the Dissociative ‘Legal Highs’ Diphenidine, Methoxphenidine and Analogues. PLOS ONE 11(6): e0157021. https://doi.org/10.1371/journal.pone.0157021.
- Wink CS, Michely JA, Jacobsen-Bauer A, Zapp J, Maurer HH. Diphenidine, a new psychoactive substance: metabolic fate elucidated with rat urine and human liver preparations and detectability in urine using GC-MS, LC-MSn , and LC-HR-MSn. Drug Test Anal. 2016 Oct;8(10):1005-1014. doi: 10.1002/dta.1946. Epub 2016 Jan 26. PMID: 26811026.
- Gerace E, Bovetto E, Corcia DD, Vincenti M, Salomone A. A Case of Nonfatal Intoxication Associated with the Recreational use of Diphenidine. J Forensic Sci. 2017 Jul;62(4):1107-1111. doi: 10.1111/1556-4029.13355. Epub 2017 Jun 9. PMID: 28597920.
Recap of Laureen Marinetti NAME & SOFT 2025 Presentations
by Denise Purdie Andrews | Dec 8, 2025 | Announcements, Drug Classes
In October 2025, Laboratory Director and Chief toxicologist Laureen Marinetti, PhD, made presentations at both the National Association of Medical Examiners 2025 Conference in Louisville, Kentucky, and at the Society of Forensic Toxicologists 2025 Conference in Portland, Oregon.
NAME 2025 Recap #1: Propoxyphene
At this year’s NAME meeting, Laureen Marinetti presented Propoxyphene was taken off the Market Fifteen Years Ago, and Yet the Lab is still Confirming it in Toxicology Casework. This presentation was developed in conjuction with her fellow Axis toxicologists, Kevin Shanks and Stuart Kurtz.
Dextro-propoxyphene was introduced for clinical use in 1963 as an opioid pain reliever for mild to moderate pain. It was sold under various names as a single-ingredient product (e.g., Darvon or Wygesic), or as part of a combination product which could contain acetaminophen, aspirin, phenacetin, and/or caffeine (e.g., Darvocet, Darvon Compound-65). The most frequent side effects of propoxyphene include lightheadedness, dizziness, sedation, nausea, and vomiting. However as the drug continued to be used it was discovered that, even during proper therapeutic use, it was cardio-toxic. Taken at therapeutic doses, there were significant changes to the electrical activity of the heart: prolonged PR interval, widened QRS complex and prolonged QT interval. In November of 2010 the FDA released a drug safety communication that recommended against the continued use of dextro-propoxyphene. Levo-propoxyphene (FDA approved in 1962) was available as an antitussive (Novrad) with no opioid like effects, however it was removed from the market in the 1970s. Eight cases confirmed positive for propoxyphene and/or norpropoxyphene in cases received by the lab from January 2020 to April of 2025. The eight cases were from the following States; Arizona (2), Florida (2), Kentucky (1), Michigan (1), and Ohio (2). Since this presentation there has been at least one additional case confirmed. Of these 8 cases there was only one in which propoxyphene may have played a role in the cause of death. This data shows that old drugs should never be counted out. Somehow they have a way of making an appearance just when you thought you would probably never see them again.
References
- Wilsa J. Raymonvil, George Hime, and Diane Moore, “Propoxyphene – Gone but not Forgotten”, Poster 27 presented at the Society of Forensic Sciences annual meeting in St. Louis, MO., 2024.
- Baselt, Randall C., Disposition of Toxic Drugs and Chemicals in Man, 12th Edition, Biomedical Publications, 2020.
NAME 2025 Recap #2: Drug Screening of Vitreous Fluid
Laureen Marinetti presented Case Comparisons of Blood, Vitreous Fluid, and Urine (Where Available): Update: Drug Screen Findings in Over 200 Cases. This presentation was also developed in conjuction with her fellow Axis toxicologists, Kevin Shanks and Stuart Kurtz.
Vitreous fluid is located in the eye between the retina and the lens. It is made up of ~ 99% water but 2 to 4 times more viscous. Vitreous fluid is commonly tested for electrolytes, glucose, creatinine, urea nitrogen, heroin exposure, recent cocaine use, and to help determine the absorption state of ethanol. It can also be tested as a second specimen to confirm drug findings from another matrix. Depending upon the circumstances of a death, vitreous fluid may be the best or only specimen available for toxicology testing. Data regarding the presence and concentration of drugs in vitreous fluid as compared to whole blood is limited. Drug penetration in to vitreous fluid depends on various factors: blood concentration, physicochemical and pharmacological properties, distribution volume, protein binding and blood retinal barrier (BRB) permeability. Drugs may diffuse passively or be actively transported across the BRB. Confirmed drug findings in over 1000 cases (more cases were added after the abstract was turned in to NAME) were compared between blood, vitreous fluid, and urine, where available. These data are important to help determine if a specific drug would be expected to be detected in vitreous fluid. Knowledge of the limitations of using vitreous fluid as a matrix in which to perform general drug screening is necessary for the proper interpretation of the results. Commonly encountered drugs did confirm in vitreous fluid with the exception of cannabinoids. In fact, a lingering opioid death may be able to be determined by comparing blood and vitreous fluid concentrations. Cases involving fentanyl will were presented as examples. Vitreous fluid can be useful in the detection of some drugs but not all drugs. In an acute overdose, drugs may not have had enough time to pass into the vitreous fluid before death occurred, an example of such a case will be presented. Also drugs at lower concentrations in blood may not be detectable in vitreous fluid, as well as those drugs that are lipophilic like benzodiazepines. Vitreous fluid has its place in toxicology testing, however due to its’ limitations, using it as a specimen for general drug screening should be avoided.
References
- Erin B. Divito, Jedediah I. Bondy, Zachary J. DiPerna, Frederick W. Fochtman, and Christopher B. Divito. A comparison of vitreous fluid and blood matrices in postmortem drug analysis, Jrn. of Analytical Toxicol., 2025, Vol 49, 351-357.
- Fabien Be´valot, Nathalie Cartiser, Charline Bottinelli, Laurent Fanton, and Je´roˆme Guitton, Vitreous humor analysis for the detection of xenobiotics in forensic toxicology: a review, Forensic Toxicol., 2015.
- Anna Pelander, Johanna Ristimaa, and Ilkka Ojanperä, Vitreous Humor as an Alternative Matrix for Comprehensive Drug Screening in PostmortemToxicology by Liquid Chromatography–Time-of-Flight Mass Spectrometry, Jrn. of Analytical Toxicol., 2010, Vol 34, 312-318.
SOFT 2025 Recap: GHB
Laureen Marinetti presented Gamma Hydroxybutyrate: What Does the Concentration Mean? Review of Ante-mortem and Postmortem Casework from 2020 – 2025. Laureen J. Marinetti, Ph.D, F-ABFT, Kevin G. Shanks, M.S., D-ABFT-FT, and Stuart A. K. Kurtz, M.S., D-ABFT-FT. The abstract (S-58) is available upon request.
Gamma hydroxybutyrate (GHB) is an endogenous molecule, a prescription drug, an illicit drug, and can be formed both ante-mortem and postmortem, making interpretation difficult. As an endogenous molecule, GHB acts as a neuromodulator with primary activity at the gamma- aminobutyrate B (GABAB) receptor; it is also a minor metabolite of GABA. Clinically, GHB was first used in the 1960s as an adjunct to anesthesia but was unpredictable regarding its duration of effect, likely due to its steep dose response curve. GHB is now used clinically to treat narcolepsy and alcohol withdrawal syndrome. As an illicit drug, GHB can be derived from its precursors, gamma butyrolactone (GBL), and 1,4-butanediol (1,4-BD), both are industrial solvents. GHB can be elevated in blood samples collected in sodium citrate tubes, and GHB can increase postmortem during the decomposition process.
The data reviewed consisted of a total of 402 ante-mortem and postmortem cases sent to the lab that were tested for GHB, either as directed testing or as part of a drug panel. GHB testing in postmortem cases resulted in 20 positive bloods, and 1 positive urine sample, out of 31 cases that were directed for GHB testing. There was a total of 9 ante-mortem blood and urine samples positive for GHB out of a total of 371 cases (2.4%) tested in the drug facilitated assault panel. Selections of these cases were presented.
The Axis cases in which GHB use was suspected were not always the cases with the highest GHB concentration. Review of the literature shows various cut offs proposed for postmortem blood and urine GHB to try to differentiate between endogenous and exogenous GHB. The cut offs vary from 20 to 50 mcg/mL in blood, and 10 to 30 mcg/mL in urine. In the Axis postmortem cases there were 7 blood samples with a GHB of less than 20 mcg/mL, 2 cases less than 30 mcg/mL, and no cases greater than 30 and less than 50 mcg/mL. The concentration range of GHB for all postmortem samples was 6.0 to 1668 mcg/mL. The endogenous GHB cut offs for ante-mortem blood and urine commonly used are 2, and 10 mcg/mL, respectively. In the five Axis ante-mortem blood cases there was a GHB concentration median of 74 and a mean of 59.7 mcg/mL, with a range of 6 to 78.2 mcg/mL. In the four Axis ante-mortem urine positive GHB cases, there were two cases less than 10 mcg/mL, a median of 8.7 and a mean of 11.7 mcg/mL, with a range of 7.5 to 248 mcg/mL.
In a 2025 study of 525 cases where no exogenous GHB use was suspected, 85% of the cases showed evidence of GHB formation postmortem, with a urine/blood ratio median of 0.52. Postmortem GHB in blood was usually less than 50 mcg/mL, but not always, 30% were above 50 mcg/mL. It was proposed that a urine/blood ratio of greater than 2.0 suggested exogenous use of GHB, however 15% of these cases had blood GHB of less than 50 mcg/mL. It was also suggested that a longer post mortem interval (PMI) resulted in a greater chance of GHB formation postmortem (Arnes et. al. 2025).
Interpretation of GHB in postmortem blood can be problematic. A detailed history and the ability to test additional specimens such as urine and vitreous fluid may be necessary.
References
- Marit Arnes, Hilde Marie Eroy Edvardsen, Line Berge Holmen, Lena KIristoffersen, and Gudrun Hoiseth, Post-mortem formation of GHB: A retrospective study of forensic autopsies, Forensic Science International 2025, 372.
- Jennifer L. Smith, Shaun Greene, David McCutcheon, Courtney Weber, Ellie Kotkis, Jessamine Soderstrom, et.al., A multicentre case series of analytically confirmed gamma-hydroxybutyrate intoxications in Western Australian emergency departments: Pre-hospital circumstances, co-detections and clinical outcomes, Drug and Alcohol Review, 2024; 1-13.
- A.W. Jones, A. Holmgren F.C., Kugelberg, and F.P. Busardò, Relationship Between Postmortem Urine and Blood Concentrations of GHB Furnishes Useful Information to Help Interpret Drug
- Intoxication Deaths, Journal of Analytical Toxicol, 2018, Vol 42, 587-591.