By Kevin Shanks, D-ABFT-FT
Mitragyna speciosa is a tree or shrub that grows in southeast Asia, particularly Thailand and Malaysia. The plant is locally known as kratom or biak-biak. It exists in the Rubiaceae family of plants, which includes the genera Coffea or caffeine-containing plants, with the most-widely known species being Coffea arabica and Coffea canephora (coffee plants). In regions of Asia, the plant has been used by either chewing the leaves or brewing them into a liquid beverage such as a tea. The leaves can also be pulverized and fashioned into a powder and then smoked or consumed orally in a capsule.

Mitragyna speciosa
Image by Ahmad Fuad Morad (CC BY-SA 2.0)
Mitragyna contains the alkaloids, mitragynine and 7-hydroxymitragynine. Approximately 60% of the plant’s alkaloid content is mitragynine and 7-hydroxymitragynine makes up about 2% of the overall alkaloid content. In lower dosages, the alkaloids produce stimulant-type effects, but at larger dosages, both compounds function as mu opioid receptor agonists. Mitragynine is considered to be approximately 13 times more potent than morphine as an analgesic, but 7-hydroxymitragynine is considered to be approximately 4 times more potent than mitragynine. 7-hydroxymitragynine is also a product of mitragynine biotransformation in the human body, thus mitragynine can be considered a prodrug for 7-hydroxymitragine. The alkaloids have also been shown to have other effects such as the blocking of serotonergic receptors and inhibition of CYP1A2, CYP2D6, and CYP3A4 enzymes.
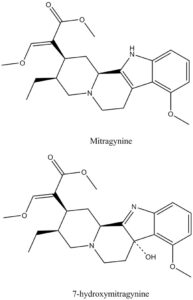
Chemical structures of Mitragynine and 7-hydroxymitragynine
Structure drawn by Kevin G. Shanks (2022)
An interesting pharmacological characteristic of mitragynine and 7-hydroxymitragynine is that when binding to opioid receptors, they exhibit biased agonism. Normally, when an opioid binds to an opioid receptor, the β-arrestin pathway is initiated – the β-arrestin pathway is responsible for most of the respiratory depression and sedation observed in opioid use and overdose. There exists evidence that shows mitragynine and 7-hydroxymitragynine do not initiate this pathway.
The United States Federal government moved to control mitragynine and 7-hydroxymitragynine as Schedule I controlled substances in 2016-2018, but backed off the legislation after public comment on the matter. They remain uncontrolled at the Federal level, but some states have passed legislation making them controlled substances in their locale.
Most forensic toxicology laboratories include only mitragynine in the scope of their testing and do not include the 7-hydroxymitragynine alkaloid/metabolite. Typical detection limits for the compound are 5-20 ng/mL in blood. At Axis Forensic Toxicology, mitragynine is included in the Comprehensive Panel (order code 70510) and/or as a directed confirmation test for mitragynine (order code 42090). Specific information about our testing can be found in the online test catalog.
Experts at Axis, alongside the Coconino County (Arizona) Medical Examiner’s Office, recently published a manuscript in the Journal of Analytical Toxicology titled “Two Single-Drug Fatal Intoxications by Mitragynine”. There have been many mitragynine-associated or related intoxications and fatalities reported over the last several years, but most have involved multiple drugs including other central nervous system depressants such as opioids, benzodiazepines, and ethanol. Sole intoxications with mitragynine leading to fatality are rare. In the published article, an analytical method for the detection of mitragynine by liquid chromatography with triple quadrupole mass spectrometry (LC-MS/MS) is detailed as well as presentation of two cases where mitragynine was certified as the single agent in the cause of death of an individual. To request a copy of this new manuscript, please contact us at [email protected].
References
Opioids. Principles of Forensic Toxicology. Fourth Edition. Barry Levine. AACC, Inc. Pages 271-291. (2017).
Mitragynine. Disposition of Toxic Drugs and Chemicals in Man. Twelfth Edition. Randall C. Baselt. Biomedical Publications. Pages 1414-1415. (2020).
“Two Single-Drug Fatal Intoxications by Mitragynine” (2022) G.S. Behonick, C. Vu, L. Czarnecki, M. El-Ters, K. Shanks. J Anal Tox, DOI: https://doi.org/10/1093/jat/bkac016
