Drug Primer: 4-ANPP
By Kevin Shanks, D-ABFT-FT
This post has been updated since its original posting Apr 7, 2022.
The presence of fentanyl in the street drug supply has rapidly exploded throughout the United States since approximately 2014. Drug overdose deaths have increased as well over the last several years and topped 100,000 deaths in the USA in 2021, with the major driving factor being fentanyl.
Fentanyl Trends. DEA Annual Report, 2020. NFLIS.
We discussed fentanyl in a previous blog post, but briefly, fentanyl is a mu opioid receptor agonist and is metabolized in the human body by the cytochrome P450 enzyme system, primarily CYP3A4, into various products. It can be dealkylated, hydroxylated, methylated, and hydrolyzed.
In the modern forensic toxicology laboratory, we monitor for the presence of unchanged fentanyl, alongside its primary metabolite, norfentanyl, in blood and urine. But, over the last several years, laboratories have added a third substance to their scope of analysis for fentanyl – 4-ANPP.
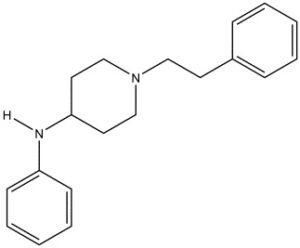
Chemical structure of 4-ANPP
Drawn by Kevin G. Shanks (2022)
4-ANPP, also known as N-phenyl-1-(2-phenylethyl)-4-piperidinamine or despropionyl fentanyl, is formed via amide hydrolysis. It is a minor metabolite of fentanyl, but it is also a precursor or starting material used in the synthesis of illicitly manufactured fentanyl and various related fentanyl analogs. 4-ANPP is reacted with propionyl anhydride to form fentanyl or some other reagent to form a related fentanyl analog such as acetylfentanyl (acetic anhydride) or cyclopropylfentanyl (cyclopropane carbonyl chloride).
Pharmacologically, 4-ANPP is inactive – it does not produce any specific effect on the body. Ultimately, its presence is merely a marker for fentanyl use or exposure. Detection of this substance in the body is highly dependent on the dose and purity of the product consumed by the individual. The toxicology alone cannot determine if 4-ANPP is present due to metabolism or if it was ingested by using an impure illicit product. There is consensus among pathologists and medical examiners to not include 4-ANPP in cause of death because of its pharmacological inactivity.
As of January 22, 2024, Axis Forensic Toxicology screens for the presence of 4-ANPP in our Comprehensive Panel with Analyte Assurance™ and reflexively confirms it with our Fentanyl & Metabolites Panel (order code 40410), which is completed by liquid chromatography with triple quadrupole mass spectrometry (LC-MS/MS). The reporting limit is 0.1 ng/mL and the substance is reported as qualitatively positive or negative.
If you have any questions regarding the presence or absence of 4-ANPP or its role in your toxicology casework, please reach out to Axis’ subject matter experts at [email protected].
References
Fentanyl. Disposition of Toxic Drugs and Chemicals in Man. Twelfth Edition. Randall C. Baselt. Biomedical Publications. Pages 844-847. (2020).
Opioids. Principles of Forensic Toxicology. Fourth Edition. Barry Levine. AACC, Inc. Pages 271-291 (2017).
2020 Annual Drug Report. National Forensic Laboratory Information System (NFLIS). Drug Enforcement Administration. Springfield, VA. NFLIS-Drug 2020 Annual Report (usdoj.gov). (Accessed March 20, 2022).
Labroo, R.B., Paine, M.F., Thummel, K.E., Kharasch, E.D. (1997) Fentanyl Metabolism by Human Hepatic and Intestinal Cytochrome P450 3A4: Implications for Interindividual Variability in Disposition, Efficacy, and Drug Interactions. Drug Metabolism and Disposition, 25: 9. 1072-1080.
Drug Primer: Fentanyl (2021). Axis Forensic Toxicology Blog. Drug Primer: Fentanyl – Axis Forensic Toxicology (axisfortox.com).
- Published in Drug Classes
Drug Primer: Fentanyl
Fentanyl was originally synthesized by Paul Janssen in 1960 and was initially marketed as Sublimaze® and used as a general anesthetic. In the mid-1990s, fentanyl was introduced to the pharmaceutical market as a transdermal patch and marketed as Duragesic®. The Actiq® transmucosal lollipop and Fentora® buccal tablet were introduced in the 2000s. Historically, fentanyl has been used to treat breakthrough pain and is used in pre-operation procedures as an analgesic and anesthetic. Fentanyl is considered a Schedule II controlled substance in the USA and is only available via physician’s prescription as a pharmaceutical.
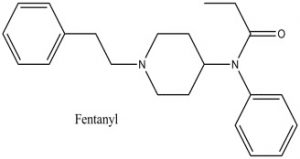
Chemical structure of Fentanyl
Structure drawn by Kevin G. Shanks (2021)
The substance is a mu (µ) opioid agonist and is considered to be 100-200 times more potent than morphine and up to 40 times more potent that diacetylmorphine (heroin) as an analgesic. Fentanyl’s blood elimination half-life is 3-30 hours but is dependent on the route of administration. Its volume of distribution is 2.5-3.5 L/kg. Fentanyl is biotransformed to its primary metabolite, norfentanyl, via the cytochrome P450 enzyme system. Other metabolites include hydroxyfentanyl, hydroxynorfentanyl, and despropionylfentanyl (4-ANPP). Effects of fentanyl use are analgesia, drowsiness, dizziness, incoordination, weakness, and lethargy. Adverse effects in overdose are central nervous system depression, respiratory depression, seizure, hypotension, apnea, hypoxia, and death.
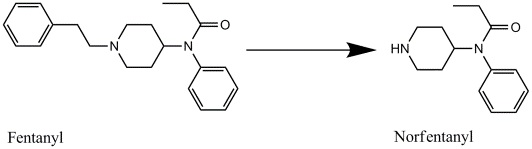
Metabolism of Fentanyl to Norfentanyl
Drawn by Kevin G. Shanks (2021)
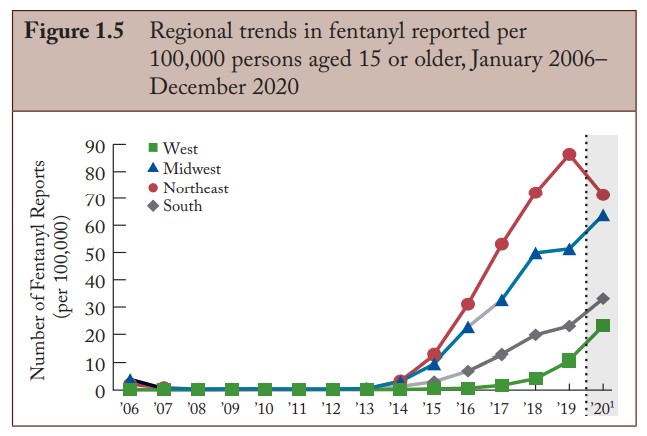
Fentanyl appeared on the illicit drug market in the USA in the 1970s. Illicitly manufactured fentanyl (of a non-pharmaceutical origin) typically originates from China and other Asian countries and can also be ordered off the “dark web” – internet sites designed to peddle illicit materials. As fentanyl has become a common adulterant in street heroin, the Drug Enforcement Administration (DEA) has reported an explosion of fentanyl-related drug seizures in recent years. From 2010 to 2019, fentanyl detections increased by 16,990%. Since 2019, the numbers have grown larger.
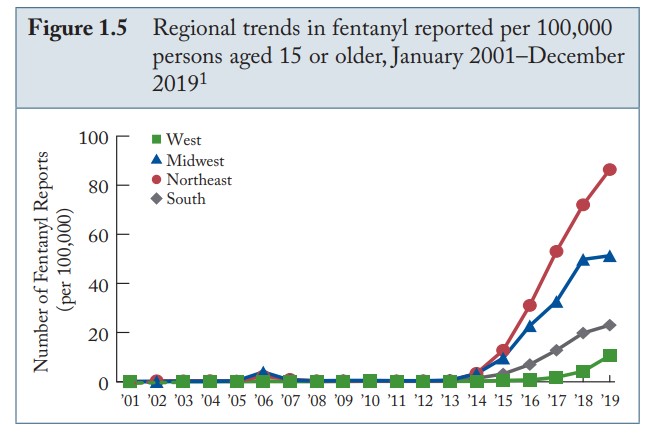
Regional trends in fentanyl 2001 – 2019.
NFLIS Annual Drug Report, 2019.
The modern forensic toxicology laboratory monitors both fentanyl and norfentanyl in blood and urine specimens. Typical detection limits for both parent drug and metabolite in biological matrices are typically 0.1 – 0.5 ng/mL. The current scope of testing and reporting limits offered by Axis Forensic Toxicology can be found in the online test catalog https://axisfortox.com/test_catalog/.
References
Fentanyl. Disposition of Toxic Drugs and Chemicals in Man. Twelfth Edition. Randall C. Baselt. Biomedical Publications. Pages 844-847. (2020).
Opioids. Principles of Forensic Toxicology. Fourth Edition. Barry Levine. AACC, Inc. Pages 271-291 (2017).
National Forensic Laboratory Information System (NFLIS). Drug Enforcement Administration. Springfield, VA. https://www.deadiversion.usdoj.gov/nflis/index.html. (accessed April 15, 2021).
NFLIS Brief: Fentanyl, 2001-2015. U.S. Department of Justice, Drug Enforcement Administration – National Forensic Laboratory Information System (NFLIS). Springfield, VA. (2017).
- Published in Drug Classes
Drug Primer: Marijuana
by Kevin Shanks, M.S., D-ABFT-FT
Marijuana, also known as cannabis, is a genus of annual flowering plants in the family Cannabaceae, and consists of the species, Cannabis sativa and Cannabis indica. The plants are native to Eastern Asia, but are cultivated all over the world. Cannabis is considered a Schedule I controlled substance by the United States Federal government, but it is legalized for medical use in 36 states and the District of Columbia (DC) and for recreational use in 18 states and DC.

Marijuana plant.
‘Legal Colorado Marijuana Grow” by Brett Levin Photography is licensed under CC BY 2.0.
The plant contains more than 500 different compounds. At least 113 of the compounds are classified as cannabinoids. The major cannabinoid is Delta-9-tetrahydrocannibinol (THC), but others include tetrahydrocannabinolic acid (THCA), cannabidiol (CBD), cannabigerol (CBG), cannabinol (CBN), and tetrahydrocannabinolic acid (THCA). Other compounds in the plant include terpenes, such as alpha-pinene, limonene, linalool, and myrcene.
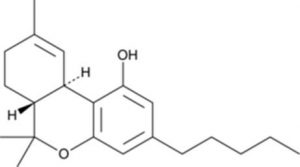
Chemical Structure of Delta-9-THC
Structure drawn by Kevin G. Shanks (2021)
THC binds to the endocannabinoid system in the body. Cannabinoid receptor 1 (CB1) is primarily located in the brain and spinal cord, while cannabinoid receptor 2 (CB2) is found in the gastrointestinal system, the immune system, and the peripheral nervous system. THC binds to these receptors and acts as a partial agonist, which means it activates them, but only has partial ability to produce a maximal response.
THC’s effects will vary according to the dose consumed, the potency of the substance, the route of administration, and the individual’s use history with the substance. When inhaled via smoking the plant or vaping THC oil, effects typically occur within minutes and last for a few hours. If taken orally via an edible such as THC-infused gummy candy and cookies, effects normally take 30-90 minutes to occur and last for 4-6 hours. Effects on the body include drowsiness, relaxation, relaxed inhibitions, altered time, altered perception, impaired learning and memory, difficulty in concentration and thought formation, and euphoria. Physiological effects include dry mouth, increased appetite, red eyes, and tachycardia.

Cannabis Edibles.
“THC-Infused Gummies” by THCProductPhotos is licensed under CC BY-ND 2.0
THC is metabolized in the liver primarily by the CYP2C9, CYP2C19, CYP2D6, and CYP3A4 enzymes to hundreds of detectable metabolites, with the main metabolites being the pharmacologically active 11-hydroxy-THC (11-OH-THC) and the pharmacologically inactive 11-nor-9-carboxy-THC (THC-COOH). THC-COOH is further conjugated with glucuronic acid and then excreted out of the body via the feces and urine.
Detection windows for the metabolite can be quite extensive and will vary according to the dose used, duration of use, and the individual using the substance. Frequent users of THC could have detectable levels of THC-COOH in their urine for up to 30 days or longer after cessation of use. Most infrequent users eliminate the metabolite within a few days up to a week after use.
A forensic toxicology lab can test for THC using many different types of instrument platforms. The initial screening test can be an immunoassay test, but can also be completed via gas chromatography with mass spectrometry (GC-MS), liquid chromatography with high resolution mass spectrometry (LC-QToF-MS) or liquid chromatography with triple quadrupole mass spectrometry (LC-MS/MS). Confirmatory testing is usually completed by either GC-MS or LC-MS/MS. In blood, both parent THC and, at minimum, the THC-COOH metabolite is monitored. Normal reporting limits for blood testing are 0.5-1 ng/mL for THC and 1-10 ng/mL for THC-COOH. In urine, labs typically only monitor THC-COOH with reporting limits for positive determination varying widely (5-300 ng/mL). The current scope of testing and screening and confirmation reporting limits offered by Axis Forensic Toxicology can be found in the online test catalog.
References
- Tetrahydrocannabinol. Disposition of Toxic Drugs and Chemicals in Man. Twelfth Edition. Randall C. Baselt. Biomedical Publications. Pages 2041-2045. (2020).
- Cannabis. Principles of Forensic Toxicology. Fifth Edition. Marilyn A. Huestis, Barry S. Levine, Sarah Kerrigan. Springer Nature Switzerland AG. Pages 389-448 (2020).
- Published in Drug Classes
Synthetic Cannabinoid Receptor Agonists (2020 – 2021)
by Kevin Shanks, M.S., D-ABFT-FT
Note: This is an excerpt of a presentation given to the LTG (London Toxicology Group) in June 2021.

Packages of SCRA products
Photograph by Kevin G. Shanks (2015)
Synthetic cannabinoid receptor agonists (SCRA) are laboratory synthesized chemicals which bind to cannabinoid receptors in the human body. There are thousands of these compounds in existence and there is no way to discuss all of them, but since 2009, many of these compounds have been sold as ingredients in smoking blends and herbal incense/potpourri in the United States. Over the last several years, the government has enacted various pieces of legislation controlling these compounds as Schedule I controlled substances and, currently, there are 43 SCRA considered to be controlled substances. The prevalence of specific synthetic cannabinoids vary over time and new compounds routinely emerge and disappear.
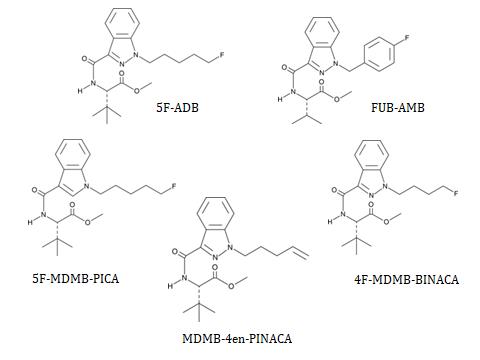
Chemical structures of 5 Common SCRA
Structures drawn by Kevin G. Shanks (2021)
According to data from the Center for Substance Abuse Research (CESAR) via quarterly Emerging Threats Reports (ETR), the prevalence of 5F-ADB and FUB-AMB have waned over the last couple of years, and the most prevalent compounds reported in 2020 were 5F-MDMB-PICA, followed by the MDMB-4en-PINACA and 4F-MDMB-BINACA. If you look at data from the National Forensic Laboratory Information System (NFLIS), compiled by the Drug Enforcement Administration (DEA) via non-biological evidence seizure, for 2020, the most common SCRAs were 5F-MDMB-PICA, MDMB-4en-PINACA, and 4F-MDMB-BINACA.
Detection of SCRA in Blood at Axis, 2018
Data compiled by Kevin G. Shanks (2021)
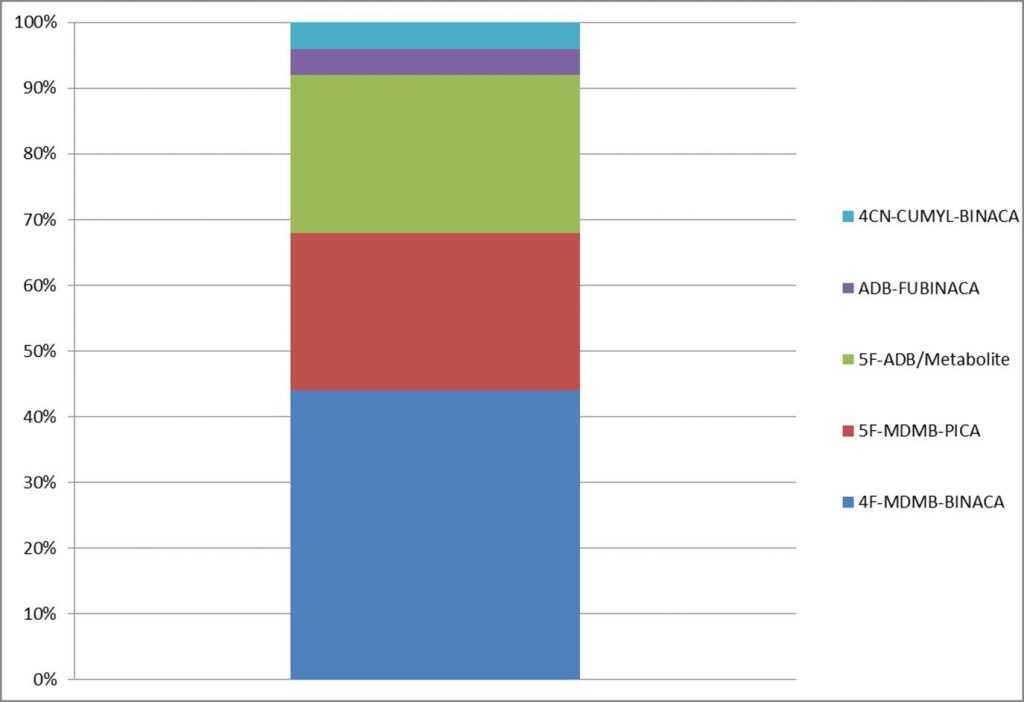
Detection of SCRA in Blood at Axis, 2019
Data compiled by Kevin G. Shanks (2021)
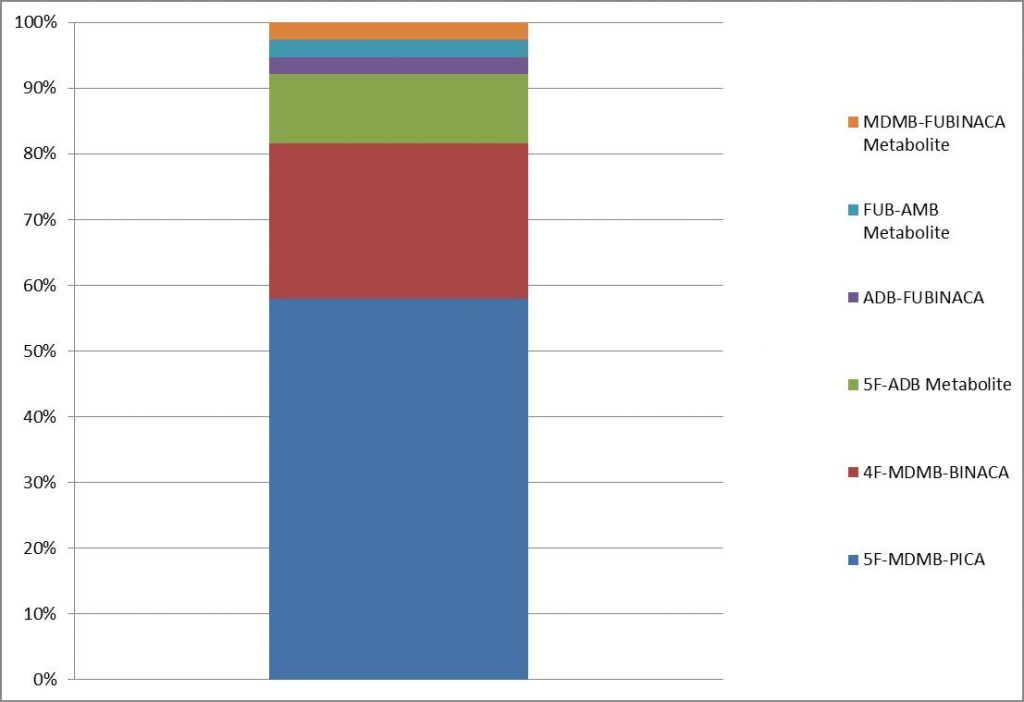
Detection of SCRA in Blood at Axis, 2020
Data compiled by Kevin G. Shanks (2021)
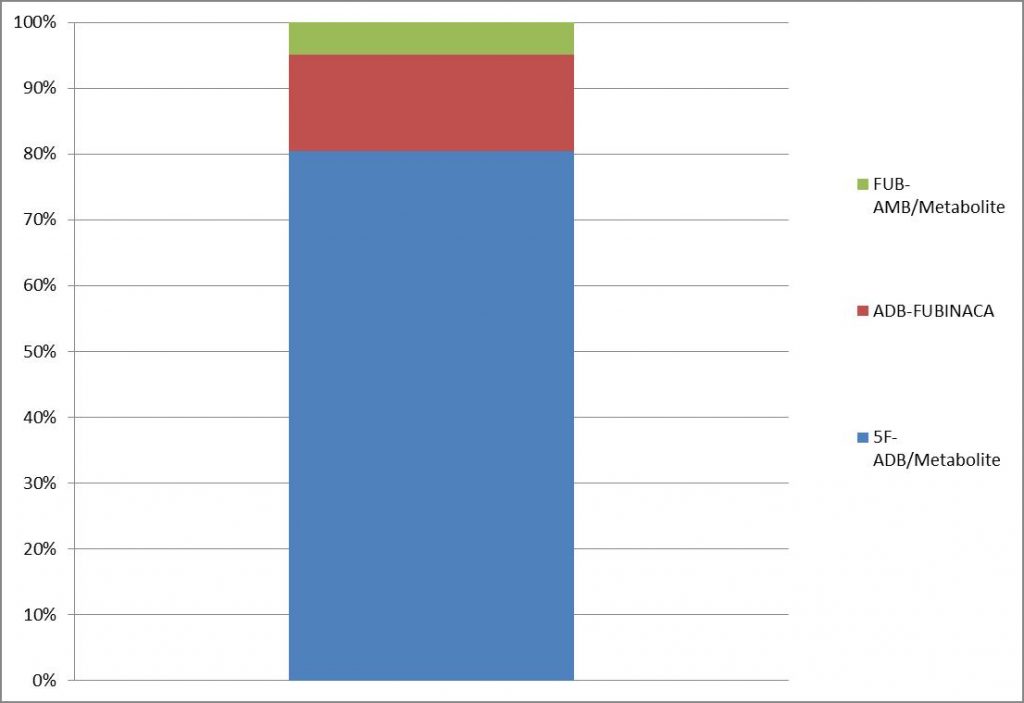
If we look at Axis Forensic Toxicology from the last few years, we can see that our data for postmortem toxicology mirrors what is reported by ETR and NFLIS. In 2018, 5F-ADB was the most prevalent SCRA (80.4% positivity in all sample tested for SCRA), while in 2019, 4F-MDMB-BINACA was the most prevalent compound (42.3% positivity). During 2020, the most prevalent compound reported in postmortem toxicology casework for synthetic cannabinoids testing was 5F-MDMB-PICA (60.0% positivity). During 2020, three compounds (5F-MDMB-PICA, 4F-MDMB-BINACA, and 5-ADB) accounted for 90+% of positive synthetic cannabinoid detections in blood samples tested.
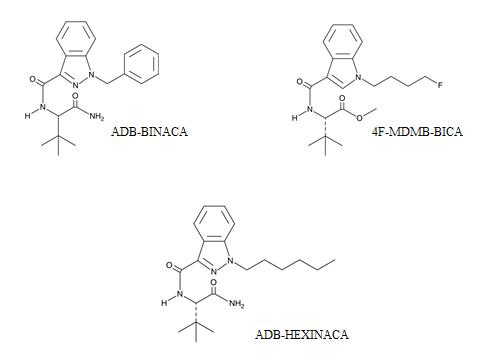
Chemical structures of 3 Emerging SCRA
Structures drawn by Kevin G. Shanks (2021)
Recognizing this ever changing scope of testing, we must always look towards the future and we need to be aware of newly reported SCRA compounds. Some of these compounds include 4F-MDMB-BICA, ADB-BINACA, and ADB-HEXINACA. What more should we expect in the future? We should expect more of the same. The beat goes on and new compounds emerge. They become prevalent. They disappear. And then new compounds take the place of the old ones.
The Puzzle Pieces of an SCRA
Image drawn by Kevin G. Shanks (2021)
Axis Forensic Toxicology continues to adapt SCRA testing to provide a relevant scope of analysis and aid in the medical-legal investigation of death and poisoning. For more information, the current scope of testing offered by Axis Forensic Toxicology can be found in the online test catalog (Order Code 42130, Synthetic Cannabinoid Panel, Blood).
If you would like a full copy of this presentation, please contact [email protected].
References
- Synthetic Cannabinoid Receptor Agonists (2020 – 2021). K. Shanks. London Toxicology Group (LTG) Virtual Meeting. (2021).
- Synthetic Cannabinoids. Disposition of Toxic Drugs and Chemicals in Man. Twelfth Edition. Randall C. Baselt. Biomedical Publications. Pages 1979-1986. (2020).
- Synthetic Cannabinoid Receptor Agonists. Novel Psychoactive Substances: Classification, Pharmacology, and Toxicology. Paul Dargan and David Wood. Academic Press (Elsevier). 317-338. (2013).
- Axis Forensic Toxicology. Laboratory Data. Indianapolis, IN. (accessed July 2021).
- Published in Drug Classes
Drug Primer: Synthetic Cannabinoids
by Kevin Shanks, M.S., D-ABFT-FT
Synthetic cannabinoids are laboratory synthesized chemicals which bind to cannabinoid receptors in the human body. The JWH series of cannabinoids was developed by Dr. John Huffman at Clemson University. UR-144 is an Abbott Labs compound which was developed during investigational drug research. ADB-FUBINACA is a synthetic cannabinoid which was synthesized by Pfizer. Numerous synthetic cannabinoids, such as 4F-MDMB-BINACA and 5F-MDMB-PICA, exist, which have no formal history in academic or pharmaceutical industry research.
In the USA, since 2008-2009, synthetic cannabinoids have been sold as ingredients in herbal incense, potpourri, or smoking blends. Throughout the last decade, various waves of legislation have been passed by the Federal government classifying several synthetic cannabinoids as Schedule I controlled substances. There are currently 43 synthetic cannabinoid substances that are controlled at the Federal level in the USA. These substances have no recognized medicinal application. Each state has its own controlled substance laws and may vary according to their regional drug trends.
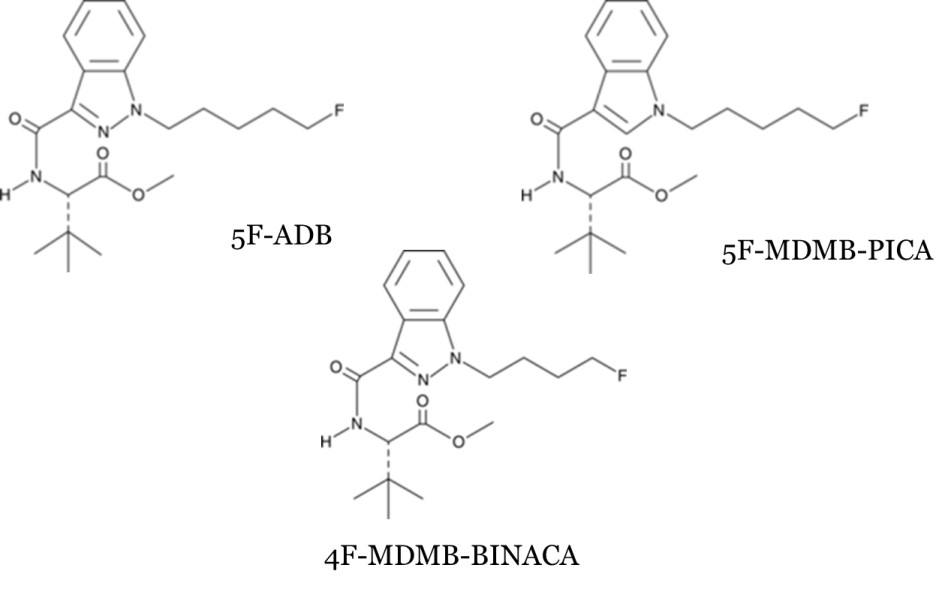
Chemical structures of 3 Synthetic Cannabinoids
Structure drawn by Kevin G. Shanks (2021)
Synthetic cannabinoids are cannabinoid receptor agonists. The cannabinoid receptor 1 (CB1) is found primarily in the central nervous system and is responsible for mediating the psychoactive effects of cannabis and related-substances. Cannabinoid receptor 2 (CB2) is located in the peripheral nervous system, the spleen, and the immune system, and is thought to be involved in pain perception mediation and immunosuppression. Unlike delta-9-tetahydrocannabinol (THC), which is a partial agonist of CB1 and CB2 receptors, the majority of synthetic cannabinoids are considered to be full agonists of the receptors.
Pharmacokinetics such as blood elimination half-life and volumes of distribution are not known for these compounds. Most synthetic cannabinoids are biotransformed to hydroxylated or carboxylic acid metabolites. Reported effects of synthetic cannabinoid use or exposure include poor coordination, sedation, slurred speech, nausea, vomiting, tachycardia, hypertension, hyperthermia, agitation, delusions, paranoia, hallucinations, psychosis, and acute kidney injury. Fatalities have occurred after the use of these substances.
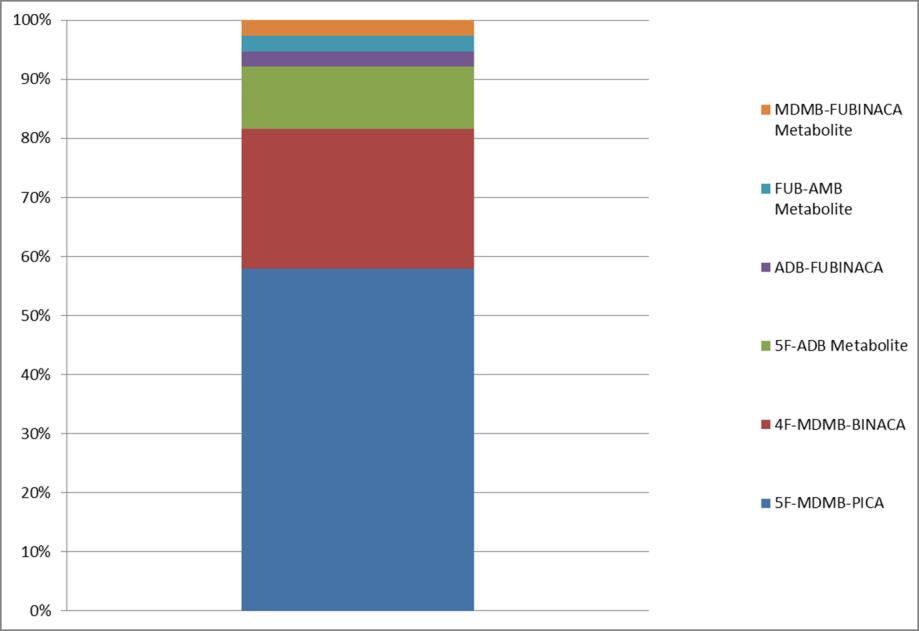
Detection of Synthetic Cannabinoids in Blood at Axis Forensic Toxicology, 2020.
Data Compiled by Kevin G. Shanks (2020).
As the prevalence of specific synthetic cannabinoids vary over time, the modern forensic toxicology laboratory should have a relevant scope of analysis. Typically only parent drugs are monitored in blood specimens, but some compounds require a metabolite be monitored in lieu of the parent drug. Detection limits for both parent drug and metabolite vary in biological matrices, but are generally 0.1 – 2 ng/mL. During 2020, three compounds (5F-MDMB-PICA, 4F-MDMB-BINACA, and 5-ADB) accounted for 90+% of positive synthetic cannabinoid detections in blood samples tested.
There current scope of testing offered by Axis Forensic Toxicology can be found in the online test catalog (Order Code 42130, Synthetic Cannabinoid Panel, Blood), and these compounds are detected by the Comprehensive Panel with Analyte Assurance™ (70510).
Axis Forensic Toxicology prides itself on its expertise in the field of novel psychoactive substances. Here are citations for papers published by Axis on the topic of synthetic cannabinoids:
- “Three Cases of Fatal Acrylfentanyl Toxicity in the United States and a Review of the Literature”, D.C. Butler, K. Shanks, G. Behonick, D. Smith, S.E. Presnell, L.M. Tormos. J Anal Tox. 42, e6-e11 (2018).
- “Synthetic Cannabinoid Product Surveillance by LC/ToF in 2013-2015”. K. Shanks, G. Behonick. J Forensic Toxicol Pharmacol, 4:3 (2016)
- “Death After Use of the Synthetic Cannabinoid 5F-AMB”. K. Shanks, G. Behonick. For Sci Int, 262, e21-e24 (2016)
- “Death Associated with the Use of the Synthetic Cannabinoid ADB-FUBINACA”. K. Shanks, W. Clark, G. Behonick. J Anal Tox, 40:3, 24-242 (2016)
- “Case Reports of Synthetic Cannabinoid XLR-11 Associated Fatalities”. K. Shanks, D. Winston, J. Heidingsfelder, G. Behonick. For Sci Int, 252, e6-e9 (2015)
- “Four Postmortem Case Reports with Quantitative Detection of the Synthetic Cannabinoid, 5F-PB-22”. G. Behonick, K. Shanks, D. Firchau, G. Mathur, C. Lynch, M. Nashelsky, D. Jaskierny, C. Meroueh. J Anal Tox, 38, 559-562 (2014)
- “Identification of Novel Third Generation Synthetic Cannabinoids in Products by Ultra Performance Liquid Chromatography and Time of Flight Mass Spectrometry”. K. Shanks, T. Dahn, G. Behonick, A. Terrell. J Anal Tox, 37: 517-525 (2013)
- “Detection of Synthetic Cannabinoids and Synthetic Stimulants in First and Second Generation Legal Highs by Ultra Performance Liquid Chromatography with Time of Flight Mass Spectrometry (UPLC/ToF)”. K. Shanks, T. Dahn, G. Behonick, A. Terrell. J Anal Tox, 36: 360-371 (2012)
- “Detection of JWH-018 and JWH-073 by UPLC/MS/MS in postmortem whole blood casework”. K. Shanks, T. Dahn, A. Terrell. J Anal Tox, 36: 145-152 (2012)
If you would like a copy of these papers, please contact [email protected].
References
- Synthetic Cannabinoids. Disposition of Toxic Drugs and Chemicals in Man. Twelfth Edition. Randall C. Baselt. Biomedical Publications. Pages 1979-1986. (2020).
- Tetrahydrocannabinol. Disposition of Toxic Drugs and Chemicals in Man. Twelfth Edition. Randall C. Baselt. Biomedical Publications. Pages 2041-2045. (2020).
- Synthetic Cannabinoid Receptor Agonists. Novel Psychoactive Substances: Classification, Pharmacology, and Toxicology. Paul Dargan and David Wood. Academic Press (Elsevier). 317-338. 2013).
- Axis Forensic Toxicology. Laboratory Data. Indianapolis, IN. (accessed April 23, 2021).
- Published in Drug Classes
