by Kevin Shanks, M.S., D-ABFT-FT
Synthetic cannabinoids are laboratory synthesized chemicals which bind to cannabinoid receptors in the human body. The JWH series of cannabinoids was developed by Dr. John Huffman at Clemson University. UR-144 is an Abbott Labs compound which was developed during investigational drug research. ADB-FUBINACA is a synthetic cannabinoid which was synthesized by Pfizer. Numerous synthetic cannabinoids, such as 4F-MDMB-BINACA and 5F-MDMB-PICA, exist, which have no formal history in academic or pharmaceutical industry research.
In the USA, since 2008-2009, synthetic cannabinoids have been sold as ingredients in herbal incense, potpourri, or smoking blends. Throughout the last decade, various waves of legislation have been passed by the Federal government classifying several synthetic cannabinoids as Schedule I controlled substances. There are currently 43 synthetic cannabinoid substances that are controlled at the Federal level in the USA. These substances have no recognized medicinal application. Each state has its own controlled substance laws and may vary according to their regional drug trends.
Chemical structures of 3 Synthetic Cannabinoids
Structure drawn by Kevin G. Shanks (2021)
Synthetic cannabinoids are cannabinoid receptor agonists. The cannabinoid receptor 1 (CB1) is found primarily in the central nervous system and is responsible for mediating the psychoactive effects of cannabis and related-substances. Cannabinoid receptor 2 (CB2) is located in the peripheral nervous system, the spleen, and the immune system, and is thought to be involved in pain perception mediation and immunosuppression. Unlike delta-9-tetahydrocannabinol (THC), which is a partial agonist of CB1 and CB2 receptors, the majority of synthetic cannabinoids are considered to be full agonists of the receptors.
Pharmacokinetics such as blood elimination half-life and volumes of distribution are not known for these compounds. Most synthetic cannabinoids are biotransformed to hydroxylated or carboxylic acid metabolites. Reported effects of synthetic cannabinoid use or exposure include poor coordination, sedation, slurred speech, nausea, vomiting, tachycardia, hypertension, hyperthermia, agitation, delusions, paranoia, hallucinations, psychosis, and acute kidney injury. Fatalities have occurred after the use of these substances.
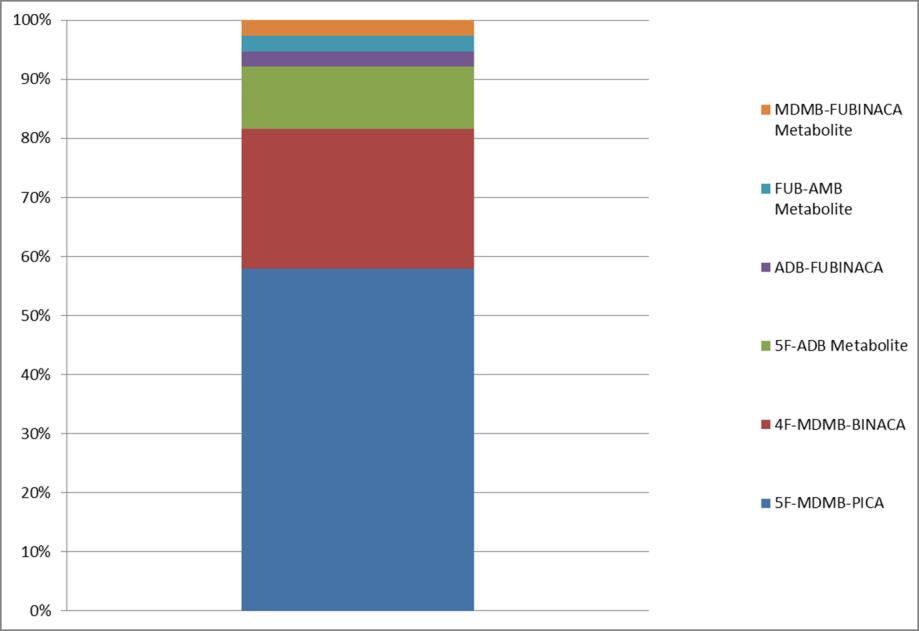
Detection of Synthetic Cannabinoids in Blood at Axis Forensic Toxicology, 2020.
Data Compiled by Kevin G. Shanks (2020).
As the prevalence of specific synthetic cannabinoids vary over time, the modern forensic toxicology laboratory should have a relevant scope of analysis. Typically only parent drugs are monitored in blood specimens, but some compounds require a metabolite be monitored in lieu of the parent drug. Detection limits for both parent drug and metabolite vary in biological matrices, but are generally 0.1 – 2 ng/mL. During 2020, three compounds (5F-MDMB-PICA, 4F-MDMB-BINACA, and 5-ADB) accounted for 90+% of positive synthetic cannabinoid detections in blood samples tested.
There current scope of testing offered by Axis Forensic Toxicology can be found in the online test catalog (Order Code 42130, Synthetic Cannabinoid Panel, Blood), and these compounds are detected by the Comprehensive Panel with Analyte Assurance™ (70510).
Axis Forensic Toxicology prides itself on its expertise in the field of novel psychoactive substances. Here are citations for papers published by Axis on the topic of synthetic cannabinoids:
- “Three Cases of Fatal Acrylfentanyl Toxicity in the United States and a Review of the Literature”, D.C. Butler, K. Shanks, G. Behonick, D. Smith, S.E. Presnell, L.M. Tormos. J Anal Tox. 42, e6-e11 (2018).
- “Synthetic Cannabinoid Product Surveillance by LC/ToF in 2013-2015”. K. Shanks, G. Behonick. J Forensic Toxicol Pharmacol, 4:3 (2016)
- “Death After Use of the Synthetic Cannabinoid 5F-AMB”. K. Shanks, G. Behonick. For Sci Int, 262, e21-e24 (2016)
- “Death Associated with the Use of the Synthetic Cannabinoid ADB-FUBINACA”. K. Shanks, W. Clark, G. Behonick. J Anal Tox, 40:3, 24-242 (2016)
- “Case Reports of Synthetic Cannabinoid XLR-11 Associated Fatalities”. K. Shanks, D. Winston, J. Heidingsfelder, G. Behonick. For Sci Int, 252, e6-e9 (2015)
- “Four Postmortem Case Reports with Quantitative Detection of the Synthetic Cannabinoid, 5F-PB-22”. G. Behonick, K. Shanks, D. Firchau, G. Mathur, C. Lynch, M. Nashelsky, D. Jaskierny, C. Meroueh. J Anal Tox, 38, 559-562 (2014)
- “Identification of Novel Third Generation Synthetic Cannabinoids in Products by Ultra Performance Liquid Chromatography and Time of Flight Mass Spectrometry”. K. Shanks, T. Dahn, G. Behonick, A. Terrell. J Anal Tox, 37: 517-525 (2013)
- “Detection of Synthetic Cannabinoids and Synthetic Stimulants in First and Second Generation Legal Highs by Ultra Performance Liquid Chromatography with Time of Flight Mass Spectrometry (UPLC/ToF)”. K. Shanks, T. Dahn, G. Behonick, A. Terrell. J Anal Tox, 36: 360-371 (2012)
- “Detection of JWH-018 and JWH-073 by UPLC/MS/MS in postmortem whole blood casework”. K. Shanks, T. Dahn, A. Terrell. J Anal Tox, 36: 145-152 (2012)
If you would like a copy of these papers, please contact [email protected].
References
- Synthetic Cannabinoids. Disposition of Toxic Drugs and Chemicals in Man. Twelfth Edition. Randall C. Baselt. Biomedical Publications. Pages 1979-1986. (2020).
- Tetrahydrocannabinol. Disposition of Toxic Drugs and Chemicals in Man. Twelfth Edition. Randall C. Baselt. Biomedical Publications. Pages 2041-2045. (2020).
- Synthetic Cannabinoid Receptor Agonists. Novel Psychoactive Substances: Classification, Pharmacology, and Toxicology. Paul Dargan and David Wood. Academic Press (Elsevier). 317-338. 2013).
- Axis Forensic Toxicology. Laboratory Data. Indianapolis, IN. (accessed April 23, 2021).
