Kevin Shanks presented on Fluorofentanyl at the Society of Forensic Toxicologists (SOFT) annual meeting in Cleveland, OH. The title of the talk is below and the abstract is available upon request.
Fluorofentanyl Detection by LC-QToFMS & Prevalence in Postmortem Toxicology
K.G. Shanks*, Stuart A.K. Kurtz, and George S. Behonick
Axis Forensic Toxicology
Fluorofentanyl is the prominent fentanyl analog that has stuck around in post-mortem casework since its resurgence in 2020. It was one of the compounds first synthesized by Janssen Pharmaceutica and has popped in and out of the illicit drug market but has never been as persistent as it is now. Our lab first started looking for fluorofentanyl in 2021.
There are 3 isomers as shown below.
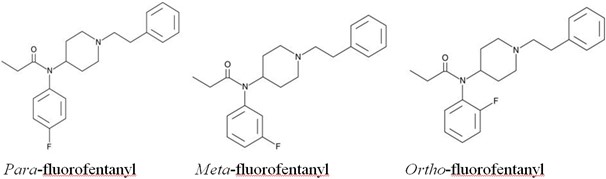
In terms of potency, they are very similar to each other. The para- and meta-fluorofentanyl isomers are about 2.5x and 5x, respectively, less potent than fentanyl. The ortho-fluorofentanyl isomer is slightly more potent than fentanyl at about 2x the potentcy.
There is some speculation as to why this particular analog has persisted. One theory is that a fluorinated precursor is being used in the synthesis process and fluorofentanyl could be a byproduct of illicitly manufactured fentanyl. Another is that the presence of fluorine could inhibit metabolism of the drug and lead to longer lasting effects. Ultimately, the answer is not clear without further information gathered.
Analysis of fluorofentanyl can be tricky. The isomers are hard to separate chromatographically and the fragmentation patterns in a mass spectrometer are nearly indistinguishable. Getting them to separate chromatographically can be beneficial to distinguish them by their retention time. There have been several methods published that have shown separation of ortho-fluorofentanyl from the para- and meta- -isomers is possible.
Given the slight difference in potency, there is some merit to being able to resolve the isomers chromatographically. However, the relative potencies are similar enough that we do not currently separate and identify them. We report them qualitatively positive/negative as fluorofentanyl with a note that we do not distinguish which isomer(s) is present. The method we use has slight variation for each isomer’s retention time but it is not enough to confidently determine which one is present in a sample.
In our casework, fentanyl was the most common drug that was detected with fluorofentanyl in 96.4% of cases. Methamphetamine (33%) and cocaine (27%) were also commonly found with fluorofentanyl. The most common NPS compound found with fluorofentanyl was metonitazene. Given its continued detection in post-mortem casework, it is beneficial to be looking for it.
Helland et al. (2017) Two Hospitalizations and One Death After Exposure to Ortho-Fluorofentanyl. Journal of Analytical Toxicology.
Gundersen et al. (2020) Metabolite Profiling of Ortho-, Meta-, and Para-Fluorofentanyl by Hepatocytes and High-Resolution Mass Spectrometry, Journal of Analytical Toxicology.
Papsun et al. (2020) Fluorofentanyl Identified in Forensic Casework as Wave of Fentanyl Related Substances Appears in the United States. NPS Discovery – Public Alert.
Krotulski et al. (2021) Examining the Evidence on Fluorofentanyl – Multidisciplinary Evaluation of this Emerging Drug with a Focus on Forensic Toxicology Investigations. SOFT 2021, S-019.
Truver et al. (2021) Identification and Quantitation of Fluorofentanyl in Postmortem Blood. SOFT 2021, P-069.
Truver et al. (2022) Toxicological Analysis of Fluorofentanyl Isomers in Postmortem Blood, Journal of Analytical Toxicology.
Trecki et al. (2022) Notes from the Field: Increased Incidence of Fentanyl-Related Deaths Involving Para-Fluorofentanyl or Metonitazene – Knox County, Tennessee, November 2020-August 2021. Morbidity and Mortality Weekly Report.
Bitting et al. (2022) Notes from the Field: Overdose Deaths Involving Para-Fluorofentanyl – United States, July 2020-June 2021. Morbidity and Mortality Weekly Report.
